Unfortunately, war is the daily media lead. We get the numbers of dead and injured for each conflict. You might consider those just injured as ‘lucky’ if you’re like me. Au contraire. Familiar issues are traumatic brain and spine injuries. Alongside lost limbs and wounds caused by bomb debris and emotional symptoms such as PTSD and depression that may persist for years, sometimes a lifetime. (Xaigham.com)
For life-saving technologies, war has unfortunately become a growth sector. I take no pleasure in saying that.
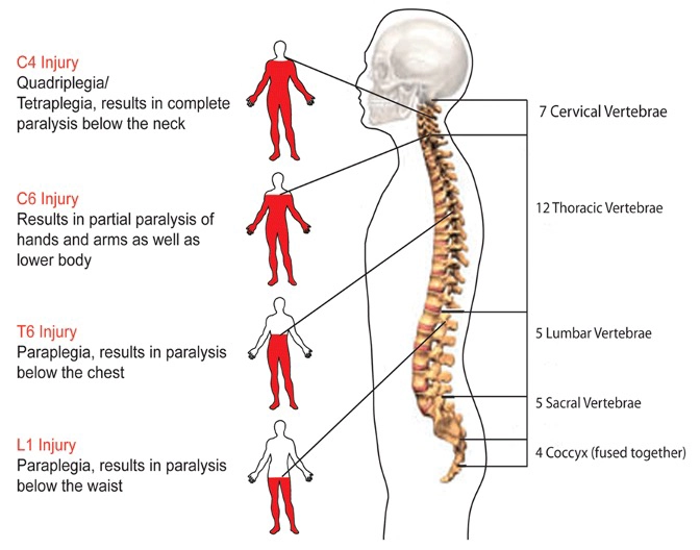
A sudden, traumatic blow to the spine (tSCI) can fracture, dislocate, crush or compress one or more of the vertebrae. A gunshot or knife wound that penetrates and cuts the spinal cord also can cause a spinal cord injury. Additional damage usually occurs over days or weeks.
The global Spinal Cord Trauma Treatment market was valued at US$ 2458.9 million in 2022 and is projected to reach US$ 3009.4 million by 2029, at a CAGR of 2.9% during the forecast period. The influence of COVID-19 and the Russia-Ukraine War were considered while estimating market sizes.
The current Middle East conflict was not included. Unfortunately, those projected growth numbers could rise significantly.
While I am using the wars and conflicts as examples of the growth of the traumatic injury market, it was already significant and this is just the US.
- A recent estimate of the annual incidence of traumatic spinal cord injury (CCI) is approximately 54 cases per one million people in the United States.
- The estimated number of people with tSCI living in the United States is approximately 302,000 persons, ranging from 255,000 to 383,000 persons.
- The average age at injury has increased from 29 during the 1970s to 43 since 2015.
- Traumatic brain injury (TBI) is defined as an alteration in brain function or other evidence of brain pathology caused by an external force. Examples of TBI include falls, assaults, motor vehicle accidents and sports injuries.
The question arises: how is this issue addressed? There are myriad companies, large and small, looking for answers.

TORONTO and HAIFA, Israel, Jan. 05, 2024 (GLOBE NEWSWIRE) — NurExone Biologic Inc. (TSXV: NRX) (FSE: J90) (NRX.V) (the “Company” or “NurExone”), a biopharmaceutical company developing biologically-guided exosome therapy for patients with traumatic spinal cord injuries.
How does it work? Stay with me; it’s pretty straightforward.
Part One: Active Ingredients
Exosomes: Exosomes, also known as extracellular vesicles, are nano-sized, naturally occurring particles in the body, secreted by cells. Exosomes, also known as extracellular vesicles, are nano-sized, naturally occurring particles in the body, secreted by cells. Can be administered non-invasively, intranasally
Part Two: Delivery
ExoTherapy: Exosomes, loaded with therapeutic molecules, cross the blood-brain barrier and reach cells and tissues for regeneration, rewiring and recovery.
Part Three: Effect
SiRNA-PTEN: The suggested PTEN inhibition-based therapeutic targets are nerve growth and regeneration after injury or damage, treatment of cardiac ischemia/reperfusion and associated disease, wound repair, and infertility.
The goal is to reverse this traumatic brain trauma as well as develop other health issues such as depression—no small accomplishment. The US FDA has granted NRX Orphan Drug Status.

The Orphan Drug Designation program provides orphan status to drugs and biologics for rare diseases that meet specific criteria. Orphan drug designation provides incentives, including:
- Tax credits for qualified clinical trials
- Exemption from user fees
- Potential for seven years of market exclusivity after approval
“Orphan-drug designation is expected to streamline our go-to-market, shorten our regulatory process, save the Company millions of dollars, and provide valuable market exclusivity. We appreciate the formal recognition of the potential impact of our therapy on the lives of patients suffering from acute spinal cord injuries,” said Dr. Shaltiel, CEO of NurExone Biologic, Ltd.
The Company also holds an exclusive worldwide license from Technion and Tel Aviv University for developing and commercializing the technology.
This technology is not only promising but appears well destined for success. In their totality, the current NRX out-front therapies could bring much relief to those seriously ‘injured’ patients who live with chronic pain and myriad challenges daily.
NurExome is a cutting-edge medical technology company. While trading has been modest, it paints a positive investment picture for the previously reasons stated. Will it pop tomorrow? No. That I can guarantee.
A savvy plan would to be to approach as a dollar-cost average investment. The deeper you dig, the more potential will become apparent.
Note Hyperlinks below.
| Stock stats Jan 5 2024 | |
| 52 Week Range | 0.1000 0.4200 |
| Volume | 7,000 |
| Avg. Volume | 4,511 |
| Market Cap | 14.475M |
| Beta (5Y Monthly) | N/A |
| PE Ratio (TTM) | N/A |
| EPS (TTM) | -0.1100 |
| Earnings Date | N/A |
| Forward Dividend & Yield | N/A (N/A) |
| Ex-Dividend Date | N/A |
| 1y Target Est | 4.01 |
| In-depth Corporate Presentation Litchfield Research |
September 5
Marc CHALLANDE
You might have ever heard that investing in pink sheets is risky for many reasons. The most famous one is because of limited information, or you can be stuck in your position because there aren’t any buyers. But this index also offers excellent opportunities if you are patient and know where to find bargain companies. In our case, Pharmagreen Biotech (OTC: PHBI) is typically the pink stock that could give a significant return on investment if you are patient enough.

The Advantages of Pink Sheets Companies
This index gathers a broad range of diverse companies. You can find companies that used to have been traded on the NASDAQ or NYSE and have subsequently been beaten down because they wouldn’t meet specific requirements. Another type of company represented on this index is the very-early stage company. If many of these companies are worth less than $1 per share, they could represent an excellent return for an investor because of volatility levels. So being early invested in a company might not be super exciting, but seeing the company you waited for so long to grow is. The Pink Sheets, technically known as OTC Pink, is run by the OTC Markets Group. OTC Markets Group hosts securities in three tiers: OTCQX, which has the most stringent listing requirements. In our case, Pharmagreen Biotech is part of the OTCQB group.

The Cannabis Market Keeps Growing
If many cannabis companies saw their valuation lowering because of many reasons over the last year and a half, the industry keeps growing, meaning the demand is higher and higher day after day. In 2020, the global cannabis market size was USD 20.47. If Covid hit many sectors, it had a staggering impact, with hemp witnessing a buoyant demand across all regions. According to Fortune Business Insights, the market exhibited a stellar growth of 50.92 in 2020. The market is projected to grow with a Compound Annual Growth Rate of 32% in 2021-2028 to reach USD 197.75. Worldwide, marijuana legislation gains momentum, increasing economic opportunities. Medical hemp has gained a lot of worldwide attention in recent years. Countries such as Germany, Finland, Israel, and Canada have taken bold steps in allowing medical marijuana use, owing to its therapeutic benefits.
Additionally, more than 25 states in the U.S. have changed their laws to legalize cannabis for medical use. In California and Florida, the product has been successfully used by mainstream doctors to treat AIDS, anorexia, arthritis, and cancer. The ongoing research and clinical trials for several marijuana-based medications are further expected to fuel the market’s growth in forthcoming years.
Why is Pharmagreen Unique?
Pharmagreen Biotech is an innovative company that uses revolutionary loT facilities to reduce the disease and contamination of plants. The Nevada-based company is specialized in the advanced science of tissue culture and doesn’t use conventional ways. Standard cloning techniques can produce genetic drift along with disease and bug infestation, resulting in lower product quality and consistency. The company builds state-of-the-art facilities that utilize the latest environmental, fertigation, and sanitation control technologies. By controlling every aspect of the plant’s growth (from specific feeding and fertilization times to ensuring each strain enjoys an optimized environment and lighting), the company ensures that every batch of new plantlets from the mother plant thrives, effectively reducing costs and solidifying the supply chain. Pharmagreen also focuses on its environmental footprint by using geothermal and solar power technologies. All these aspects work together consistently, reliably, and cost-effectively to produce the highest quality flower tops and biomass. The company is focused on its business development with Long Valley Farms while it completes its fundraising for the acquisition. The micro-business license application, which will enable significant and immediate revenue generation for the cannabis business, is being processed by Mendocino County, California, and various relevant governing bodies.
Peter Wojcik (CEO) leads the company. Peter Wojcik has over a decade of experience in high-valued exits across past-successful startups and research in cannabis as a therapeutic agent. He is also highly knowledgeable in the world of economics.
“We are laser-focused on getting revenues started with the lowest CAPEX as soon as possible. We plan to create products based on the whole plant and fungi, thereby maximizing the beneficial and therapeutic effects, the same approach we use with cannabis. Once the micro cannabis license gets approved, we will add cannabis-based products, both stand-alone and combined with other ingredients, to target specific ailments and help people prevent illness first.”
Peter Wojcik, CEO of Pharmagreen Biotech
What Should You Remember?
Investing in pink sheets companies can be risky and tricky, but choosing the right ones can grant you a significant return on investment if you can afford to wait for the company to run its business entirely. This is why Pharmagreen Biotech (OTC: PHBI) is the right pick for you. Despite being an early-stage company, it should start producing any time soon and see its revenues sharply grow from $500k to $9M. The company’s technology eliminates disease and contamination, reduces growing time, and down the road, assures a higher quality product.
Marc has been involved in the Stock Market Media Industry for the last +4 years. After obtaining a college degree in engineering in France, he moved to Canada, where he created Money,eh?, a personal finance website. He then contributed to building Guerilla Capital, a Capital Markets company and FirstPhase Media where he is head of research. At10xAlerts, he writes articles and conducts interviews on many sectors, including technology, metals & mining markets.
-
Marc CHALLANDEhttps://10xalerts.com/author/marcchallande/
-
Marc CHALLANDEhttps://10xalerts.com/author/marcchallande/
-
Marc CHALLANDEhttps://10xalerts.com/author/marcchallande/
-
Marc CHALLANDEhttps://10xalerts.com/author/marcchallande/
All Articles
The latest
3 Small-cap Gold Juniors to Take Notice of $ELEM $GLDR $SOMA
Golden Rapture – The Most Undervalued Gold Exploration Company trading in the Junior Markets (CSE:GLDR)
INVESTMENT: THIS AI FIRM JUST RELEASED SOME GAME-CHANGING NEWS
Subscribe
©2024 10xAlerts
